IN BRIEF: Two hundred experimental drugs intended to treat Alzheimer's disease have failed in the past 30 years. Without new therapies, the number of patients worldwide will increase dramatically by 2050.
A ray of hope has come recently from a clinical trial that showed that dementia's cognitive impairment might be prevented by paying close attention to various health factors.
Participants in the study who followed a regimen of health-related steps registered improvements on cognitive measures such as memory and mental-processing speed.
Results of this research suffice for health care professionals to begin making a series of recommendations to patients on diet, exercise and levels of social engagement that may help prevent dementia.
More people are reaching a ripe old age than ever before. Life expectancy has increased from 45 years in the early 1800s to more than 80 today in most European countries and in Japan, Canada and Australia, among other nations. In fact, if the trend continues, a majority of babies born in these countries today will live past their 100th birthday.
This greater longevity comes with some bad news. Although we manage to survive longer than preceding generations did, we often gain time without being healthier in those extra years. Studies from different parts of the world indicate that after age 60, most people have at least one chronic disorder, such as heart disease or diabetes, and a recent population-based study in Sweden found that at 80, only around one out of 10 individuals were free of chronic diseases. In fact, most people older than 80 in this population had two or more chronic illnesses.
Modern medicine has become increasingly adept at treating and controlling many of these conditions, but a few common age-related diseases have defied attempts to find preventive therapies or cures—most notably, Alzheimer's disease, the leading cause of dementia. Alzheimer's follows a merciless course that progressively deprives a person of memories and a sense of self-identity, losses that have devastating consequences for relatives and friends as well.
In the U.S., around 32 percent of people older than 85 have received an Alzheimer's diagnosis, often combined with other types of dementia, such as that caused by vascular disease. Estimates put the number worldwide with any kind of dementia at about 50 million. By 2050 if no new treatments succeed in delaying the condition, more than 130 million may suffer with some form of the disease. About 60 to 70 percent of these patients will have Alzheimer's dementia, and around 20 to 25 percent will be classified as suffering from the vascular form of the illness.
In spite of more than 100 ongoing clinical trials, no cure or drug has been able to halt the course of Alzheimer's. More than 200 experimental drugs intended to treat it have failed during the past 30 years. But all is not grim. New findings from a gold-standard clinical trial the two of us have been involved in indicate that cognitive impairment can be prevented or delayed even in the absence of new drugs by promoting changes in behavior and by managing vascular risk factors.
The study we conducted took its inspiration from epidemiological research that sought ways to reduce the risk of Alzheimer's. Such investigations, called association studies, measure, at different points, health-related variables, such as depression, high blood pressure, diet and exercise. Then, usually many years later, they investigate whether individuals go on to acquire a particular disorder. A strong correlation between a variable and the disease in question suggests that some aspect of our health history might merit being labeled as a risk factor. Also, if one of the observed variables correlates with a low risk of disease, the finding can be taken as a sign that it might be protective.
HOW WE LIVE
During the past 10 to 15 years association studies have indicated that maintaining cardiovascular health and adopting certain measures—good diet, exercise, an active social life and the achievement of higher educational levels—may diminish the risk of both Alzheimer's and other dementias later in life, even for those who carry genes that place them at increased risk. Epidemiologists have also increasingly begun to discover specific factors that may be protective—for instance, living with a partner or consuming a Mediterranean diet (mainly based on fish, vegetables, fruit and olive oil). Some studies suggest that controlling, for example, blood pressure and diabetes may constitute primary prevention—protections against the onset of a disease. They might also afford secondary prevention, slowing memory loss and other symptoms in the earliest stages of illness.
Although association studies can flag a purported protective factor, they unfortunately cannot prove that taking these steps will actually prevent dementia. People who eat a Mediterranean diet or exercise three times a week may avoid disease because of some other variable ignored by epidemiological sleuths.
Epidemiologists try to deal with this problem by making statistical adjustments, but it is virtually impossible to discount every aspect of a person's life that could confound a study's conclusions. They can never be sure that they have succeeded, and sometimes the relevant data may not even exist. Obtaining reliable data on early childhood experiences is extremely difficult, although what happens during the first years might influence development of high blood pressure or some other aspect of health that contributes to the development of Alzheimer's late in life. Lacking all the necessary data can produce spurious associations among variables that lead to the wrong conclusions. Statistical equations, moreover, all but collapse under their own weight if too many variables are considered at the same time.
In 2010 the problem of establishing causality from association studies prompted a U.S. National Institutes of Health conference to conclude that not enough evidence existed to make firm recommendations on whether a given factor might diminish the risk of cognitive decline. To overcome this difficulty, a systematic review article from the NIH conference suggested that Alzheimer's researchers launch randomized controlled trials—and that any study undertaken examine not one but multiple factors that might prove to be pivotal in preventing dementia.
The randomized controlled trial is the gold standard that science uses to determine whether a treatment is in fact effective—in this case, whether a true cause-and-effect relation links variables—perhaps diet and exercise—with outcomes such as avoiding cognitive decline. Participants in these trials are randomly assigned to either a treatment or a control group. To avoid biasing results, neither the researchers nor the study participants know which individuals are assigned to a particular group.
In the past investigators have undertaken relatively few long-term randomized controlled trials of whether lifestyle changes can improve health because accurately monitoring day-to-day behavior is challenging. But the NIH conference experts still recommended them as the best way to proceed because of the need for substantive data and because past randomized controlled trials looking at only a single variable had failed or produced mixed results. Moreover, Alzheimer's researchers had come to recognize the need to learn from successes in advancing prevention strategies for heart disease and diabetes, which relied on studies that looked at multiple risk factors.
THE STUDY
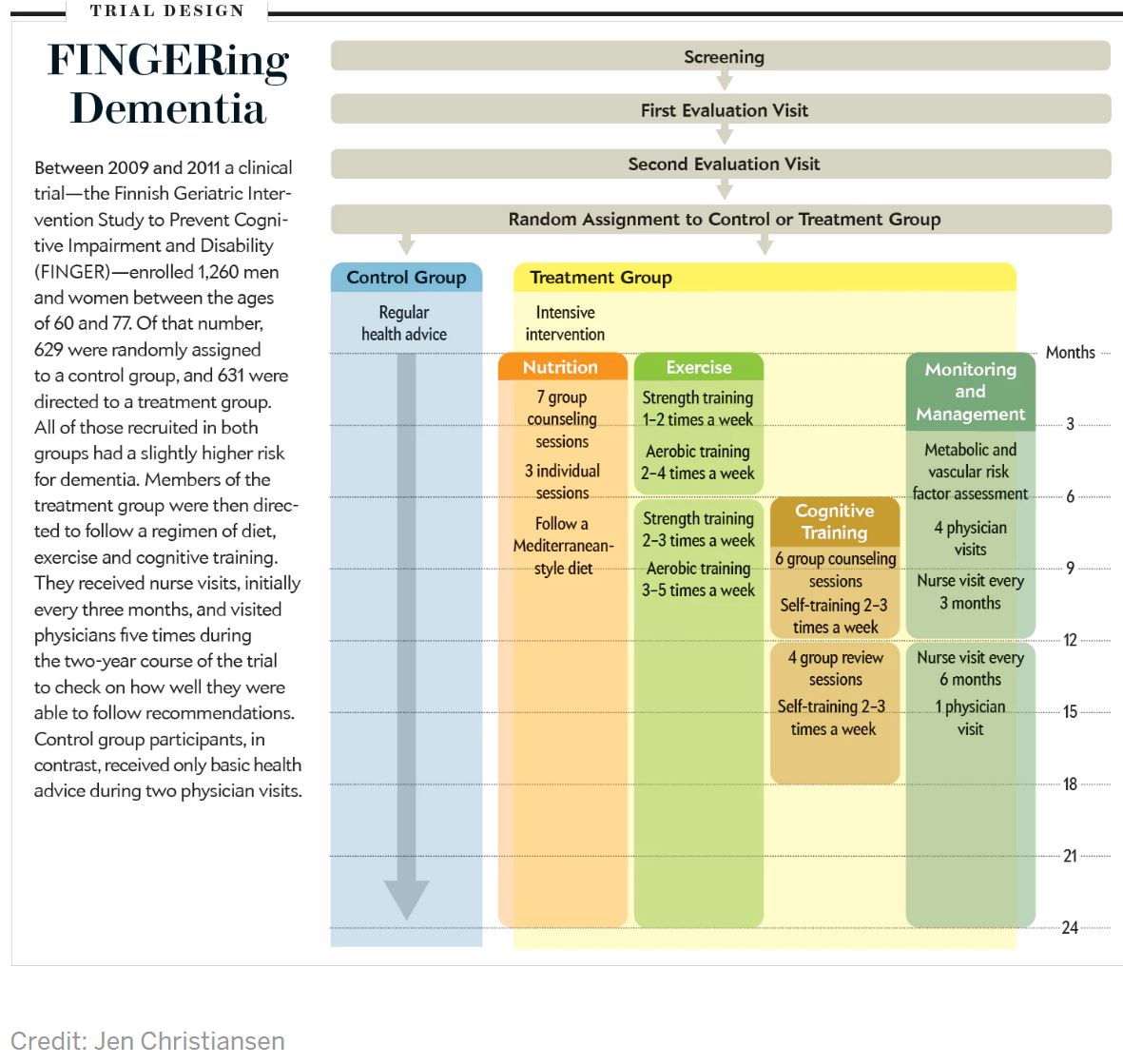
Since 2010 several long-term, randomized controlled studies have progressed, and findings are now being reported. Our project—the Finnish Geriatric Intervention Study to Prevent Cognitive Impairment and Disability (FINGER)—was the first to publish. FINGER's goal was to assess the effect on cognitive health of improved diet, physical exercise and mental training while providing regular health advice and cardiovascular health monitoring.
We and our colleagues wanted to know whether, over a two-year period, overall cognitive performance would differ between the treatment group—631 men and women between the ages of 60 and 77—and the 629 members of the control group. (The control subjects received health advice, and their cardiovascular health was checked regularly. If health problems were identified, such as high blood pressure, control group members were referred to a physician.) To optimize chances that the trial would succeed, we structured the pool of subjects to consist of individuals who had an elevated risk for cognitive decline, as judged by scores on a survey that measures dementia risk (the Cardiovascular Risk Factors, Aging and Dementia risk score).
In comparison with the control group, the intervention group received a mix of nutritional guidance, cognitive training and physical exercise, and the subjects' cardiovascular status was also more intensely monitored. The nutritional guidance aimed at a healthy balance of protein, fat, carbohydrates, dietary fibers and salt and included restrictions on consumption of trans-fatty acids, refined sugar and alcohol, all in accordance with recommendations by Finland's National Nutrition Council. The main ingredients in the recommended diet were fruits, vegetables, whole grains and rapeseed oil, along with a fish meal at least twice a week. The only food supplement was vitamin D.
STEPS TO PREVENTION: Physical activity was a key part of the FINGER intervention. Credit: Courtesy of Finnish Geriatric Intervention Study to Prevent Cognitive Impairment and Disability
Physical exercise included muscle-strength training, aerobic exercise and postural balance. The exercise program was individually tailored to each participant and led by a physiotherapist during the first six months, then performed independently by the participants in group sessions. The initial recommendation was to go to the gym one to two times a week for sessions of 30 to 45 minutes to train muscle strength. After six months of gradual increases in intensity, the participants reached a maximum level of two to three gym sessions of 60 minutes a week, which they then maintained during the remaining 18 months. In addition, subjects received advice to perform aerobic training twice a week and increase their regimen gradually to three to five times a week. Based on individual preferences, they could choose to engage in Nordic walking, aqua gym, jogging or calisthenics as part of the aerobic component of the intervention.
The intervention group also used a computer program to train on different cognitive tasks to enhance executive function (planning and organizing), memory improvement and mental speed. After six introductory group lessons led by a psychologist, group members trained on their own two to three times a week in 10- to 15-minute sessions during two six-month periods. Four group reviews monitored progress along the way and discussed topics such as age-related cognitive changes.
The participants also had regular checkups of metabolic and vascular health. They met with a study nurse on six occasions to measure weight, blood pressure, and hip and waist circumference. Physicians also reviewed these and other laboratory results with subjects five times during the two-year trial period, using them as a basis to encourage changes in daily habits.
By any standard, FINGER proved to be an intensive intervention for most participants, a drastic change to their lives during the two years it lasted. That most of them adhered to the routine was regarded as a success in itself. Only 12 percent dropped out, usually citing health problems. Further, just 46 of the 631 participants in the intervention group experienced any difficulty completing the tasks—the most common adverse event was muscle pain following physical training. We concluded that a comprehensive program of changes to one's daily activities in later life is practical to implement. But the more important question was whether the goal of preserving cognition had actually been reached.
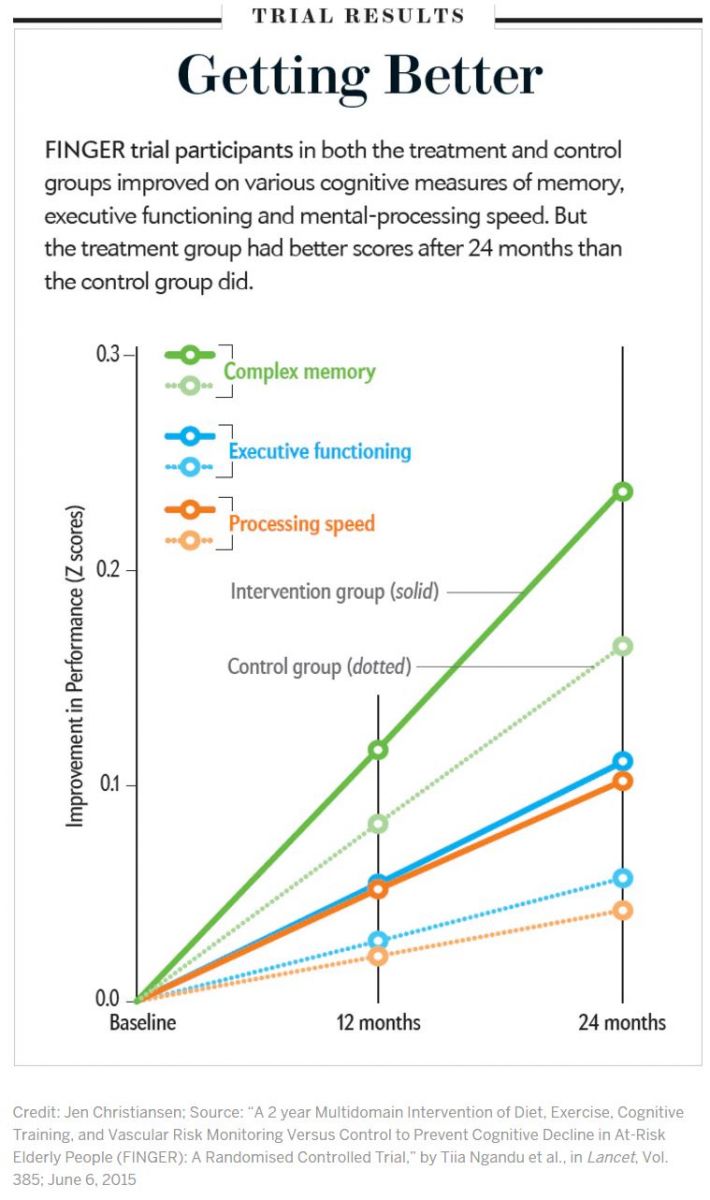
After two years the treatment group showed clear benefits: overall cognitive performance improved on average in both the treatment and control groups, but the intervention group benefited 25 percent more than did controls. Another analysis—looking at the number of people who deteriorated in cognitive performance over the two years—showed a surprising result: the risk of waning cognitive performance turned out to be 30 percent higher in the control group. Improvement in control groups often occurs in randomized controlled trials for various reasons. People usually perform better on the same tests the second time they take them. But FINGER did not have a control group in the traditional sense. The regular meetings with health advice and cardiovascular monitoring attended by participants in the control group became a kind of mini intervention.
Many control subjects may have been inspired by these sessions to make at least some changes to benefit cognitive functioning. Even though we knew that this arrangement might narrow the differences in results between the two groups, we also had an ethical obligation to ensure that the intervention had at least some benefit for the control group. Still, after tallying our results, we did not lose confidence that it represented a real effect, because the treatment group improved by a significantly higher degree than the control group did.
Participants in the treatment group also registered significant gains in other areas as well. They improved in specific cognitive domains that help people in daily activities that often decline with aging. The intervention group logged an 83 percent improvement over the control group in executive function, a 150 percent better score in speed of processing (time required to perform mental tasks) and a 40 percent performance increase in complex memory tasks (remembering long lists, for instance).
Delving further into our data, we found that participants with a gene variant (APOE e4) that puts them at higher risk for Alzheimer's seemed to receive somewhat more benefit from the specified changes than others without it, more proof of the intervention's effectiveness. Those in the treatment group who carried the risk gene had a slower rate of cellular aging, as measured by biological markers called telomeres, caps at the end of a chromosome.
FINGER EXTENSION
We now have fairly good evidence that a combination of improved diet, physical exercise, mental and social stimulation, and management of cardiovascular problems can improve cognition even after age 60. But we still have more follow-up work to do on our original results.
Finding an improvement in mental functioning after two years implies—but does not prove—that changing one's diet and exercise habits can protect against dementia. To investigate whether it is possible to delay the onset of dementia, we have to consider the long presymptomatic period that is typical of the various forms of dementia. Alzheimer's develops over probably 15 to 20 years before cognitive problems can be diagnosed. We might therefore need to follow people over an extensive period. Of course, it will also be necessary to decide at which point this type of study becomes too expensive and impractical to complete.
Another question we needed to address was whether someone experiencing the brain changes that precede actual cognitive complaints might be helped by counteracting the physiological alterations. Could changes to one's activities of the type implemented in FINGER push back the onset of cognitive issues? Postponing symptoms by two to five years would translate into a significant improvement in public health. Such a delay would mean that many people would probably not receive a dementia diagnosis, because they would die of other causes first.
To investigate some of these questions, we have put in place an extension of FINGER for another seven years. For this next phase, we plan to use brain scans to determine whether good habits might counteract the breakdown in connections among neurons and stem atrophy in certain brain areas, both hallmarks of Alzheimer's. Blood tests may pinpoint whether adopting behaviors that appear to improve cognition lessens inflammation, cellular stress and a deficiency of the proteins that help to ensure brain health—signs of pathology that often turn up in the postmortem brains of Alzheimer's patients.
We are also working with various research teams to bring together findings from studies similar to our own conducted in other countries. The comparisons can help determine whether our discoveries generalize to different populations—combining results can also increase the statistical power of the research and enable more detailed analyses of timely interventions. We could, for instance, compare the levels of physical training among intervention groups from the various studies to identify optimal levels to preserve brain health.
What we have learned from FINGER can also serve as a model for similar studies that attempt to mine the epidemiology literature to extrapolate multiple risk factors that can then be examined in a randomized controlled trial. We are now collaborating on two such projects: the European Union's Healthy Aging through Internet Counseling in the Elderly and the Multimodal Strategies to Promote a Healthy Brain in Aging.
It will not be necessary to wait another decade for health care professionals to start making recommendations to their patients. Already FINGER has provided enough evidence to start suggesting that patients take up the health measures we investigated. If the NIH decides to convene a new conference, it might now come to a more optimistic conclusion than the one held seven years ago that was unable to endorse any preventive steps.
The agency might also take assurances from recent reports indicating a decreasing rate of Alzheimer's in the U.S. and for all forms of dementia in both the U.S. and several European countries. This decline may stem from behavioral changes people have implemented on their own after hearing about scientific studies on such changes that can benefit cognitive health.
In the face of a multitude of drug failures, prevention may prove the best way to manage the dementia epidemic, as it has for many other chronic diseases. The take-home message from FINGER is that it may never be too early to take measures to prevent Alzheimer's—and fortunately, it may never be too late, either: changes to the way one lives seem to help some people even after cognitive decline has already begun.


Spread the word