During the first year of the COVID-19 pandemic, the “lab leak” theory gained little traction. Sure, U.S. President Donald Trump suggested SARS-CoV-2 originated in a laboratory in Wuhan, China—and called it “the China virus”—but he never presented evidence, and few in the scientific community took him seriously. In fact, early in the pandemic, a group of prominent researchers dismissed lab-origin notions as “conspiracy theories” in a letter in The Lancet. A report from a World Health Organization (WHO) “joint mission,” which sent a scientific team to China in January to explore possible origins with Chinese colleagues, described a lab accident as “extremely unlikely.”
But this spring, views began to shift. Suddenly it seemed that the lab-leak hypothesis had been too blithely dismissed. In a widely read piece, fueled by a “smoking gun” quote from a Nobel laureate, a veteran science journalist accused scientists and the mainstream media of ignoring “substantial evidence” for the scenario. The head of WHO openly pushed back against the joint mission’s conclusion, and U.S. President Joe Biden ordered the intelligence community to reassess the lab-leak possibility. Eighteen scientists, including leaders in virology and evolutionary biology, signed a letter published in Science in May that called for a more balanced appraisal of the “laboratory incident” hypothesis.
Yet behind the clamor, little had changed. No breakthrough studies have been published. The highly anticipated U.S. intelligence review, delivered to Biden on 24 August, reached no firm conclusions, but leaned toward the theory that the virus has a natural origin.
Fresh evidence that would resolve the question may not emerge anytime soon. China remains the best place to hunt for clues, but its relative openness to collaboration during the joint mission seems to have evaporated. Chinese officials have scoffed at calls from Biden and WHO Director-General Tedros Adhanom Ghebreyesus for an independent audit of key Wuhan labs, which some say should include an investigation of notebooks, computers, and freezers. Chinese vice health minister Zeng Yixin said such demands show “disrespect toward common sense and arrogance toward science.” In response to the increasing pressure, China has also blocked the “phase 2” studies outlined in the joint mission’s March report, which could reveal a natural jump between species.
Despite the impasse, many scientists say the existing evidence—including early epidemiological patterns, SARS-CoV-2’s genomic makeup, and a recent paper about animal markets in Wuhan—makes it far more probable that the virus, like many emerging pathogens, made a natural “zoonotic” jump from animals to humans.
Some of those clues have led Michael Worobey, an evolutionary biologist at the University of Arizona who has done groundbreaking work on the origins of HIV and the 1918 flu, further away from the lab-origin theory. Although he always viewed it as less likely, he co-signed the Science letter calling for a more thorough investigation of the lab-leak hypothesis. But like at least one other signatory, he now has second thoughts about that plea, in part because it heightened political tensions. “I think it probably did more harm than good in terms of actually having relevant information flow out of China,” he says.
Jesse Bloom, an evolutionary biologist at the Fred Hutchinson Cancer Research Center who spearheaded the Science letter, says the lab-origin theory will continue to thrive until the Chinese government becomes more cooperative. “I don’t think Chinese scientists are less trustworthy,” says Bloom, who has sharply criticized China for attempting to “obscure” data about early COVID-19 cases. “But it’s clear that, at least in relation to this topic, they are operating under strong constraints imposed by the government.”
AT ITS CORE, the lab-origin hypothesis rests on proximity. A novel coronavirus, genetically linked to bats, surfaced in a city that’s home to the Wuhan Institute of Virology (WIV), which has long specialized in studying bat coronaviruses, and two smaller labs that also handle those viruses. One or more lab workers could have become infected by accident, then passed the virus to others. Lab accidents are not unheard of, after all: SARS-CoV, the coronavirus that causes severe acute respiratory syndrome (SARS), has infected researchers as many as six times after the global outbreak of that disease ended in July 2003.
A researcher’s infection with SARS-CoV-2 needn’t have happened in Wuhan itself. Alina Chan, a gene therapy researcher at the Broad Institute who also co-signed the Science letter, cites a study by WIV researchers, published in 2018, that sampled blood from 218 people who lived 1000 kilometers from the city near caves that were home to coronavirus-infected bats. Six of these people had antibodies that suggested prior infections by SARS-related bat coronaviruses, a branch of the family tree that includes SARS-CoV, SARS-CoV-2, and close cousins. Wuhan researchers have visited that area repeatedly and “easily could have picked up something from a human who already carried a human-adapted form of a SARS-related virus,” Chan says.
Shi Zhengli, the lead bat coronavirus scientist at WIV, denies that anyone at the lab fell ill around the time SARS-CoV-2 emerged. In an email interview with Science in July 2020, she wrote that “all staff and students in the lab” were tested for SARS-CoV-2 and related coronaviruses and were negative.

Speculation about a lab leak has focused on the Wuhan Institute of Virology, which has studied bat coronaviruses for years. Ng Han Guan/AP Images
Still, in January, days before Trump left office, the U.S. Department of State said the “government has reason to believe that several researchers inside the WIV became sick in autumn 2019.” And on 23 May, The Wall Street Journal reported the existence of an “undisclosed U.S. Intelligence report” that said three WIV researchers “sought hospital care” in November 2019. The story had no details about their illnesses, and some have noted that Chinese hospitals provide care for all ailments, including minor ones.
Virologist Robert Garry of Tulane University finds it improbable that a Wuhan lab worker picked up SARS-CoV-2 from a bat and then brought it back to the city, sparking the pandemic. As the WIV study of people living near bat caves shows, transmission of related bat coronaviruses occurs routinely. “Why would the virus first have infected a few dozen lab researchers?” he asks. The virus may also have moved from bats into other species before jumping to humans, as happened with SARS. But again, why would it have infected a lab worker first? “There are hundreds of millions of people who come in contact with wildlife.”
Another data point argues against infected researchers playing a role, Garry says. As the WHO joint mission report spells out, clusters of early COVID-19 cases had links to multiple Wuhan markets around the same time, which Garry says supports the idea of infected animals or animal traders bringing the virus to the city. A lab worker with COVID-19 would have had to make “a beeline not just to one market, but to several different markets,” he says. “You can’t rule it out, but then why the markets? Why not a soccer game or a concert or 100 other different scenarios?”
But David Relman, a Stanford University microbiome researcher who also co-signed the Science letter, questions the “hopelessly impoverished” data on the earliest COVID-19 cases. “I just don’t think we have enough right now to say anything with great confidence,” Relman says.
Linfa Wang, a molecular virologist at the Programme in Emerging Infectious Diseases at Duke-NUS Medical School in Singapore who has collaborated extensively with WIV on bat coronavirus studies, has a simpler reason for dismissing the lab-leak hypothesis. “Accidents can only happen when you already have a live virus in culture that can leak,” Wang says. Bat coronaviruses are notoriously hard to grow. Shi told Science last year that her lab had more than 2000 bat fecal samples and anal and oral swabs that tested positive for coronaviruses. But the lab had only isolated and grown three viruses over 15 years, Shi said, and none closely resembled SARS-CoV-2. Some have questioned Shi’s veracity—she may well be under pressure from the Chinese government—and noted inconsistencies in her statements, but several scientific collaborators outside China have high regard for her integrity.
Wang also discounts reports that WIV has live bats. “Many years back” the lab conducted immune studies on live bats, Wang says, but these were not of the genus Rhinolophus—the only one found to harbor SARS-related coronaviruses—which no lab has ever been able to keep alive in captivity.
A great deal of speculation about the pandemic’s origin has centered on six men who developed severe respiratory illnesses in 2012 after clearing bat feces from a copper mine in Mojiang, in China’s Yunnan province. Three of them died. Lab-origin proponents have suggested the men were infected with a coronavirus, a belief fed by a 2013 master’s thesis that provided no direct evidence. That bat virus, they argue, either was SARS-CoV-2 or was turned into it through genetic engineering.
When the miners fell ill, Shi and co-workers were asked to sample bats at the mine, which they did on several occasions. They discovered nine new SARS-related viruses (see sidebar, below). One of these, dubbed RaTG13, is 96.2% genetically identical to SARS-CoV-2, the closest overall similarity yet found. A loose-knit group whose members call themselves DRASTIC—for the Decentralized Radical Autonomous Search Team Investigating COVID-19—has driven a heated discussion about possible links between RaTG13 and SARS-CoV-2.
Shi has reported that her lab tested blood from the miners and did not find evidence of coronaviruses or antibodies to them. Wang, who helped with these analyses, finds the assertion that the team suppressed evidence of SARS-CoV-2’s link to the Mojiang mine preposterous. “We wanted to prove that a coronavirus caused the deaths,” says Wang, who grew up in Shanghai but is now an Australian citizen. “If we proved that another SARS-like virus was in humans in China that would have been scientifically brilliant,” he says. “It’s a Science or Nature paper. No scientist is going to wait for this to leak.”
Even Bloom agrees with that logic. “That’s one of the strongest arguments you can make against a lab accident,” he says. “On the other hand, I feel like a lot of these questions could be resolved pretty easily by enhanced transparency.”
IN THE MOST ELABORATE lab-leak scenarios, SARS-CoV-2 is not a naturally occurring virus, but was created at WIV. That would bring worldwide condemnation on China, but it would also devastate the field of virology. There has been an intense debate over the past decade about the scientific value of “gain-of-function” (GOF) studies, which deliberately create pathogens that are more virulent or more transmissible to humans—or both—than their natural cousins. Some say GOF studies can help identify and thwart future threats, but critics argue the potential benefits don’t outweigh the risk of creating and unleashing pandemic pathogens.
Shi has created chimeric viruses in the past to get around the difficulty of growing coronaviruses isolated from bats. In work with Peter Daszak of the EcoHealth Alliance and Wang, described in a 2017 paper in PLOS Pathogens, WIV made chimeras using the genetic “backbone” of one of the bat coronaviruses her lab could culture and genes that coded for the surface protein, called spike, from newly found coronaviruses.
Scientists disagree about whether this was GOF research. Shi says it was not, because the hybrid viruses her group created were not expected to be more dangerous than the original strains. Anthony Fauci, head of the U.S. National Institute of Allergy and Infectious Diseases, which helped fund the study, told Congress it does not qualify as GOF research under NIAID’s guidelines. Relman finds the GOF label “vague and confusing” and instead describes this as “unnecessarily risky research.”
Definitions aside, if Shi was creating chimeric viruses, SARS-CoV-2 may have been one of them, lab-leak proponents say. They also note biosecurity measures at the lab were relaxed. In her 2020 Science interview, Shi denied conducting chimeric virus experiments beyond those reported in the 2017 paper, but she acknowledged doing some coronavirus studies in biosafety level 2 facilities. That’s one level lower than even Ralph Baric, a coronavirus researcher at the University of North Carolina, Chapel Hill, who has collaborated with Shi, thinks is appropriate. Shi stressed that the work complied with all Chinese regulations.
Still, many scientists contend that SARS-CoV-2 can’t be a lab concoction because no known virus is close enough to have served as its starting material. Some have countered that RaTG13, the virus found in the Mojiang mine, could have been that backbone. That makes no sense, asserts a “critical review” by Garry, Worobey, and 19 other scientists that Cell published online on 19 August. More than 1100 nucleotides, the building blocks of RNA, separate the genomes of the two viruses, and the differences are scattered in a way that doesn’t suggest deliberate engineering.
“Nobody has the sort of insight into viral pathogenesis to design something as really devious as SARS-CoV-2,” Garry says. Three other bat viruses more similar to SARS-CoV-2 than RaTG13 in some key genomic regions are also unlikely to have been used as a template for the pandemic virus, according to the paper.
The “smoking gun” evidence that SARS-CoV-2 was engineered, in the words of virologist and Nobel laureate David Baltimore, has not held up either. Spike has a cleavage site, a spot where a human enzyme named furin cuts the protein, which helps SARS-CoV-2 infect cells. Since early in the pandemic, lab-origin proponents have claimed that no SARS-related bat coronaviruses have this feature, leading to speculation that a lab added the site to a virus so it could infect humans. When retired New York Times writer Nicholas Wade made the case for a lab leak this spring, the furin cleavage site, buttressed by Baltimore’s provocative words, was an essential part of the argument.
But it’s dead wrong, say many coronavirus specialists and evolutionary biologists. The SARS-related coronaviruses are in the beta genus, one of four in the Coronaviridae family. Several members of that genus feature furin cleavage sites, which appear to have evolved repeatedly. And one SARS-CoV-2–related virus, described in a Current Biology paper last year by a team led by Shi Weifeng of Shandong First Medical University, has three of the four amino acids that constitute the furin cleavage site, which is “strongly suggestive of a natural zoonotic origin” for SARS-CoV-2, the authors concluded.
Baltimore has backpedaled the statement. He did not know several bat beta coronaviruses have the furin cleavage site, he acknowledged in an email to Science. “here is more to this story than I am aware of,” he wrote. “The furin cleavage is the most ridiculous stuff,” Wang says.
Instead of genetically manipulating a virus, a lab could also have created SARS-CoV-2 by passaging, a technique in which researchers grow a virus in a lab dish or an animal, harvest it, and repeat the process again and again, allowing mutations to accrue. But again, they would have needed to start with a close relative of SARS-CoV-2. There’s no evidence that this precursor existed in any lab. And passaging in cell cultures often deletes the furin cleavage site or makes viruses weaker.
Even the U.S. intelligence community during the Trump administration discounted the suggestion that SARS-CoV-2 was “manmade.” The report requested by Biden, which sought input from several groups in the intelligence community, similarly concludes that the virus “was probably not genetically engineered.” (It also said there was “broad agreement” that it “was not developed as a biological weapon.”)
THE JOINT MISSION REPORT from WHO, which runs more than 300 pages and delves into everything from the viral sequences of the earliest cases to pharmacy sales, has several little-noticed findings that make a natural origin appear more likely than a lab leak, says Kristian Andersen, an evolutionary biologist at Scripps Research who co-authored the recent Cell paper with Garry and Worobey. “It wasn’t the perfect report,” he says, but it was “a great start to a collaborative study on understanding the origin of SARS-CoV-2.”
The earliest official announcement about the pandemic came on 31 December 2019, when Wuhan’s Municipal Health Commission reported a cluster of unexplained pneumonia cases linked to the city’s Huanan seafood market. The WHO report devotes much attention to details about Huanan and other Wuhan markets, but also cautions that their role remains “unclear” because several early cases had no link to any market. But after reading the report, Andersen became more convinced that the Huanan market played a critical role.
One specific finding bolsters that case, Wang says. The report describes how scientists took many samples from floors, walls, and other surfaces at Wuhan markets and were able to culture two viruses isolated from Huanan. That shows the market was bursting with virus, Wang says: “In my career, I have never been able to isolate a coronavirus from an environmental sample.”
The report also contained a major error: It claimed there were “no verified reports of live mammals being sold around 2019” at Huanan and other markets linked to early cases. A surprising study published in June by Zhou Zhao-Min of China West Normal University and colleagues challenged that view. It found nearly 50,000 animals from 38 species, most alive, for sale at 17 shops at Huanan and three other Wuhan markets between May 2017 and November 2019. (The researchers had surveyed the markets as part of a study of a tick-borne disease afflicting animals.)
Live animals can more easily transmit a respiratory virus than meat from a butchered one, and the animals included masked palm civets, the main species that transmitted SARS-CoV to humans, and raccoon dogs, which also naturally harbored that virus and have been infected with SARS-CoV-2 in lab experiments. Minks—a species farmed for fur that has acquired SARS-CoV-2 infections from humans in many countries— were also abundant. “None of the 17 shops posted an origin certificate or quarantine certificate, so all wildlife trade was fundamentally illegal,” Zhou and his colleagues wrote in their paper. (Zhou did not respond to emails from Science.)
It’s unclear why the international members of the WHO joint mission were not told about the live market mammals by their Chinese counterparts. “I’m really disappointed that came out after [the report],” says WHO’s Maria Van Kerkhove, who acknowledges contributing to the oversight herself because she mistakenly ignored a draft of the paper that the authors sent her when they first submitted it in October 2020.
Worobey says the paper played a key role in tilting his thinking away from the lab-origin hypothesis. “The fact that early cases were linked to the market, and that the market was selling what were very likely intermediate hosts?” he says. “All of that is probably trying to tell us something.”
Worobey suspects that after a SARS-CoV-2 progenitor jumped from animals to humans, it pingponged back and forth, steadily adapting to its new host. This could have happened at the market and gone unnoticed for weeks, as the outbreak only surfaced when several people became severely ill, a relatively rare outcome of a SARS-CoV-2 infection. Or the virus could have first infected animal farmers in remote villages. “If this happened in a small town, it’s quite probable it would never have taken off,” says William Hanage, an evolutionary biologist at Harvard University. Then animal traders might have brought the virus to markets in Wuhan, a city of 11 million.
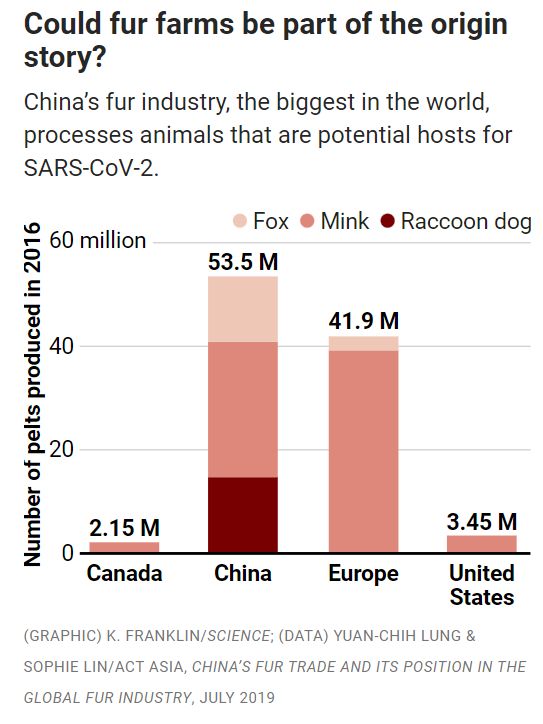
Linda Saif, a veterinarian at Ohio State University, Wooster, says China’s enormous fur industry is at the top of her list of places to hunt for SARS-CoV-2’s precursors. Saif cites a report showing the vast majority of the world’s pelts from raccoon dogs and foxes—both canids, a family readily infected with SARS-CoV-2—are from animals farmed or trapped in China (see graphic, above). The country produces half of the world’s mink pelts, too.
SO WHERE TO NOW? Bloom would like more details about the earliest human cases of COVID-19 and says WIV should share bat coronavirus sequences in a database it removed from the internet in September 2019, claiming the site had been hacked. “That could put a lot of this to rest,” he says. Sales data from Wuhan markets could help, too. If researchers could trace who farmed or trapped the live animals sold there and who delivered them to the markets, those people could be interviewed and perhaps sampled for evidence of past infections.
In a comment published by Nature on 25 August, the international members of the joint mission warned it’s time to get on with “phase 2” because the window for some studies is closing. But WHO is reconfiguring the team. It recently announced a new International Scientific Advisory Group for Origins of Novel Pathogens (SAGO) that will provide “rapid advice” to launch the follow-up studies described in the mission report, but also study the origins of future outbreaks. The Chinese government has shown no signs it will welcome SAGO members, but Wang is hopeful it will have a change of heart: “In an ideal world, we need a Chinese collaboration.”
Chinese scientists are conducting their own studies into potential natural origins, but few outside the country know details. “Unfortunately, finding out what is being done is getting harder by the day because the lab-leak stuff has turned COVID origins into a major political weapon,” says one Western researcher who asked not to be identified. “My colleagues in China are nervous and feeling great pressure.”
China has been pushing the theory that the virus came from another country—maybe brought in on frozen food, or, according to baseless propaganda, concocted at a U.S. military lab. “It’s comical,” Worobey says. “The big picture here is China is doing everything it can to push the narrative that this pandemic started outside of China.” He suspects that while rejecting the lab-leak theory, the Chinese government is also unenthusiastic about pursuing a natural origin, fearing that proof would expose China to further blame for a pandemic even if the discovery exonerated Chinese scientists. “I think at some point they thought, here’s the strategy: We try to muddy the waters,” he says.
But even without China’s cooperation, there are ways to move ahead. Some studies elsewhere have already yielded intriguing leads. Researchers have found coronaviruses in bats in neighboring countries that suggest evolutionary pathways from an ancestor of SARS-CoV-2 to the pandemic virus. More clues may come from studies in Southeast Asia of wild pangolins—the only other species to date found to harbor a close SARS-CoV-2 relative.
Researchers can also hunt for cases outside of China that predate the December 2019 outbreak. One possibility, Wang says, is to check the blood of Wuhan visitors or residents who were in the city in the months before, including the 9000 athletes from more than 100 countries who attended the Military World Games there in October 2019. (A new antibody assay from his lab, he says, can distinguish between SARS-CoV-2 and related viruses that may have preceded it.)
The search will never lead us to patient zero, the first person to be infected by SARS-CoV-2, Hanage says. “Humans are looking for a story,” he says. “They want Columbo to come in and just somehow get somebody to confess or show what actually happened.” Instead, there are “possible stories” about SARS-CoV-2’s origin—some more probable than others—and stories that can be excluded, Hanage says. “And the space of possible stories in which there was a natural origin in or around the markets is much larger than the space of possible origins in which the Wuhan Institute of Virology is involved.”
Jon Cohen is a staff writer for Science.
When you become a AAAS member, you’re not just getting a magazine about science—you’re becoming part of the community that’s driving scientific progress. Get the very best research from around the world delivered right to you weekly. Your membership also helps AAAS educate and communicate with policymakers at local, state, and national levels, bringing evidence-based research to bear on critical issues such as climate change, STEM education, and public funding of science research.


Spread the word