Sunday Science: Fossil Teeth Reveal How Brains Developed In Utero Over Millions of Years of Human Evolution
Fossilized bones help tell the story of what human beings and our predecessors were doing hundreds of thousands of years ago. But how can you learn about important parts of our ancestors’ life cycle – like pregnancy or gestation – that leave no obvious trace in the fossil record?
The large brains, relative to overall body size, that are a defining characteristic of our species make pregnancy and gestation particularly interesting to paleoanthropologists like me. Homo sapiens’ big skulls contribute to our difficult labor and delivery. But the big brains inside are what let our species really take off.
My colleagues and I especially wanted to know how fast our ancestors’ brains grew before birth. Was it comparable to fetal brain growth today? Investigating when prenatal growth and pregnancy became humanlike can help reveal when and how our ancestors’ brains became more like ours than like our ape relatives’.
To investigate the evolution of prenatal growth rates, we focused on the in-utero development of teeth – which do fossilize. By building a mathematical model using the relative lengths of molar teeth, we were able to track evolutionary changes in prenatal growth rates in the fossil record. Based on our model, it looks as if pregnancy and prenatal growth became more humanlike than chimplike almost 1 million years ago.
Pregnancy and delivery come with a lot of risks for parent and baby. Jimy Lindner/EyeEm via Getty Images
Gestation and the human brain
Pregnancy and gestation are important periods – they guide future growth and development and set the biological course for life.
But human pregnancy, and particularly labor and delivery, cost a lot of energy and are often dangerous. The large fetal brain requires a lot of nutrients during development. The rate of embryonic growth during gestation, also known as the prenatal growth rate, exacts a metabolic and physiological toll on the gestating parent. And the tight fit of the infant’s head and shoulders through the pelvic canal during delivery can lead to death, for both the mother and child.
As a trade-off to those potential downsides, there must be a really good reason to have such large heads. The justification is all the abilities that come along with having a big human brain. The evolution of our large brain contributed to our species’ dominance and is associated with increased use of technology and tools, creation of art and the ability to survive in diverse landscapes, among other advances.
The timing and sequence of events that led to the evolution of our large brains is entangled with the ability to find and process more resources, through the use of tools and cooperative group work, for example.
By investigating changes in prenatal growth, we are also investigating changes in how parents gathered food resources and distributed them to their offspring. These increasing resources would have also helped drive the evolution of an even bigger brain. Understanding more about when prenatal growth and pregnancy became humanlike at the same time reveals information about when and how our brains did too.
Humans have the highest prenatal growth rate of all primates living today, at 0.41 ounces/day (11.58 grams/day). Gorillas, for example, have a much larger adult body size than humans, but their prenatal growth rate is only 0.29 ounces/day (8.16 grams/day). Because more than a quarter of all human brain growth is completed during gestation, the rate of prenatal growth directly relates to how big an adult brain grows. How and when Homo sapiens‘ high prenatal growth rate evolved has been a mystery, until now.
What teeth can tell about prenatal growth
Researchers have spent centuries investigating variation in fossilized skeletal remains. Unfortunately brains – let alone gestation and prenatal growth rate – don’t fossilize.
The developing brain of a human being gestating at 26 weeks. Tesla Monson
But my colleagues and I started thinking about how teeth develop very, very early in utero. Your permanent adult teeth started developing long before you were born, when you were just a 20-week-old fetus. Tooth enamel is more than 95% inorganic, and the vast majority of everything we see in the vertebrate fossil record is teeth, or has teeth.
Building off this realization, we decided to investigate the relationship between prenatal growth rate, brain size and the lengths of teeth.
We measured the teeth of 608 recently living primates from skeletal collections all around the world. We compared those measurements to rates of prenatal growth that we calculated from average gestation length and mass at birth for each species. We also looked at endocranial volume – essentially how much space is inside the skull – as a proxy for brain size.
We found that the rate of prenatal growth is significantly correlated with both adult brain size and relative tooth lengths, across apes and monkeys.
Because prenatal growth is so tightly correlated with relative molar lengths, we were able to use this statistical relationship to generate a mathematical equation that predicts prenatal growth rate from teeth alone. With this equation, we can take a few molar teeth from an extinct fossil species and reconstruct exactly how fast their offspring grew during gestation.
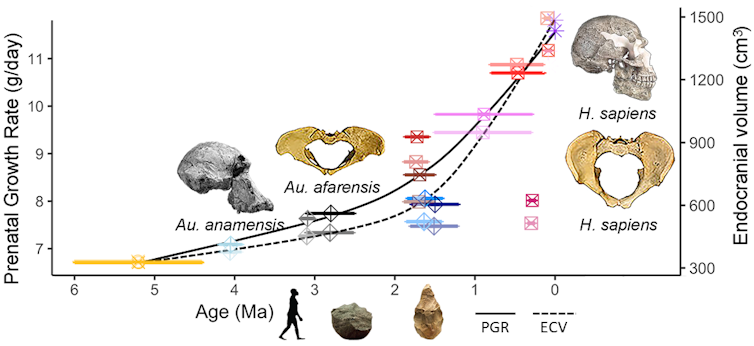
Using the new equation, researchers found that prenatal growth rates increased over millions of years of human and hominid evolution. Tesla Monson, CC BY-ND
Using our new method, we then reconstructed prenatal growth rates for 13 fossil species, building a timeline of changes over the past 6 million years of human and hominid evolution. “Hominid” describes all the species on the human side of the family tree after the split about 6 million to 8 million years ago from the common ancestor we shared with chimpanzees. From our new research, we now know that prenatal growth rates increased throughout hominid evolution, reaching a humanlike rate that exceeds what we see in all other apes less than 1 million years ago.
A fully human prenatal growth rate appeared with the evolution of our species Homo sapiens only around 200,000 years ago. But other hominid species living in the past 200,000 years, such as Neanderthals, also had “human” prenatal growth rates. Which genes were involved in these changes in growth rate remains to be investigated.
Equation means teeth now reveal even more
Even with only a few teeth and some of the jaw, a trained expert can tell countless things about an extinct individual – what species it was, what kind of diet it ate, whether it competed for mates through fighting, how old it was when it died, whether or not it had any serious health issues and more.
Now, for the first time, we can add to that list knowing what pregnancy and gestation were like for that individual and other members of its species. Teeth can even indirectly hint at the emergence of human consciousness, via evolving brain size.
Interestingly, our model suggests that prenatal growth rates started increasing well before the emergence of our Homo sapiens species. We can hypothesize that having a fast prenatal growth rate was necessary for growing that big brain and evolving human consciousness and cognitive abilities.
These are the sorts of big-picture questions this research lets us start to formulate now – all from just a few teeth.![]()
Tesla Monson, Assistant Professor of Anthropology, Western Washington University
This article is republished from The Conversation under a Creative Commons license. Read the original article.



Spread the word